The Certificate of Analysis is a legally binding document that is issued by a certification authority regarding a product. The document attests that the product has undergone extensive testing in a certified lab.
The document will include how the product follows the specifications and regulations provided by both the customer and the manufacturer as well as any governing entities.
How is the Certificate of Analysis Utilized?
The certificate can be used to reassure the customers of the quality of the product that is being sold. The Certificate can be required according to an agreement between the buyer and seller or required by governmental entities that are responsible for regulation and inspection of products during safety inspections and quality testing.
These entities include:
- Food And Drug Administration (FDA)
- International Organizations for Standardization
- Clinical Laboratory Improvement Amendments (CLIA)
- Internal Quality System Procedures
Elements of Certificate of Analysis
There is the information required within the Certificate that is needed to be covered in a comprehensive manner to ensure that the product is adequate and is complying with the regulations. The quality control or quality assurance departments develop Certificates of Analysis in a timely manner with accurate detailed information regarding the product that is coordinated with the shipping timeframe to ensure that the shipment will be accepted by the inspectors upon delivery.
The required information is explained as followed:
- The title of document: The document should contain the correct title which reads “Certificate of Analysis.” Some companies may issue other names such as the certificate of compliance, but the collect title should be the certificate of analysis.
- Name and address of the certifying body: This is the first part of the certificate whereby all the details of the certifying company are indicated. The details include; the name of the organization and the physical address as well.
- Supplier information: The content at the beginning of the certificate is the information regarding the supplier of the materials used in the manufacturing process. This may include the name of the supplier, logo, and contact information such as Email, phone number, PO box, FAX number, and full address.
- Description of the certified reference material: Information regarding the materials that are being used in the product that is being certified is required. This must include the name of the product, serial numbers, lot number, and description of the product that is being certified. It should also be taken into consideration that the records need to link the packaged products to the materials that are filled within to keep an accurate trace record as well as to ensure that the package(s) that is being shipped contains the required materials as stated in the certificate.
- Certified reference material code and batch number: Each and every certified material has its code and batch number which are different from the other certified reference materials.
- Description of certified reference material: The information described in this part includes the description of the batch number of the starting material or the raw materials used. It measures and finds out if the item is completely pure or there are any impurities detected. If there are any impurities found they should be recorded on this part.
- Intended use of certified reference material: This is the part where the reason for issuing the certified reference material is listed.
- Instructions for the CRM’s correct use: The certificate should also include the safety data sheets. The reference material should be utilized according to the conditions given and in the sequence outlined in the document.
- Hazardous situation: The document should contain the material safety data sheet that lists the nature of any hazards and the precautions to take so as to avoid the dangers.
- The certified values and their uncertainties: In the case of any error in the budget, it should be recorded on the risk budget. All CRM should have a stated uncertainty that should be defined on the certificate.
- Conformance information: Evidence of conformance is the most important information within the document. This information will explain how the product has been inspected by a certified lab or quality assurance department and whether the materials used within the product are conforming to the specifications provided by the manufacturer, government regulations as well as any specifications requested by the customer or not. A standard performance specification is usually provided as a form of comparison to ensure the quality of the products up to a certain standard. It will include the data of lab tests, methods used, special characteristics of the product, and the regulation that were implemented on the products according to the standard of the industry as:
- Values obtained by individual laboratories or methods: In the case where another lab is asked to check the results of the project carried out, the information should be reported on the certificate as well.
- Uncertified values: The values are listed to provide information and should be used as a reference.
- The level of homogeneity: There is a section that consists of the data about the identity of the item.
- Trace-ability: The results of the measured property which can relate to the stated reference should be indicated.
- Date of certification: This is the part where the first dates of the certificate of reference material and the certificate are stated.
- Period of validity: The part indicates the date that the CRM’s and certificate are expected to expire.
- Transportation information: Information regarding the transportation of the products will include the name and address of the customer, date of shipping, purchase order billing, the bill of lading number, and destination. It should also include the transporting railcar number or trailer number as it helps the receiver to confirm the materials that are being delivered. This will aid in keeping an accurate record of each shipment.
- Further information: In the case where there is additional information to be added, it should be indicated as well.
- Names and signatures of certifying officers: The name of the official who takes responsibility for the information of the certified analysis must be included on the certificate.
Certificate of Analysis Examples
Here are a few samples of the certificate of analysis.
Model certificate of analysis requirements

Certificate of analysis template for food

Certificate of analysis template for chemical
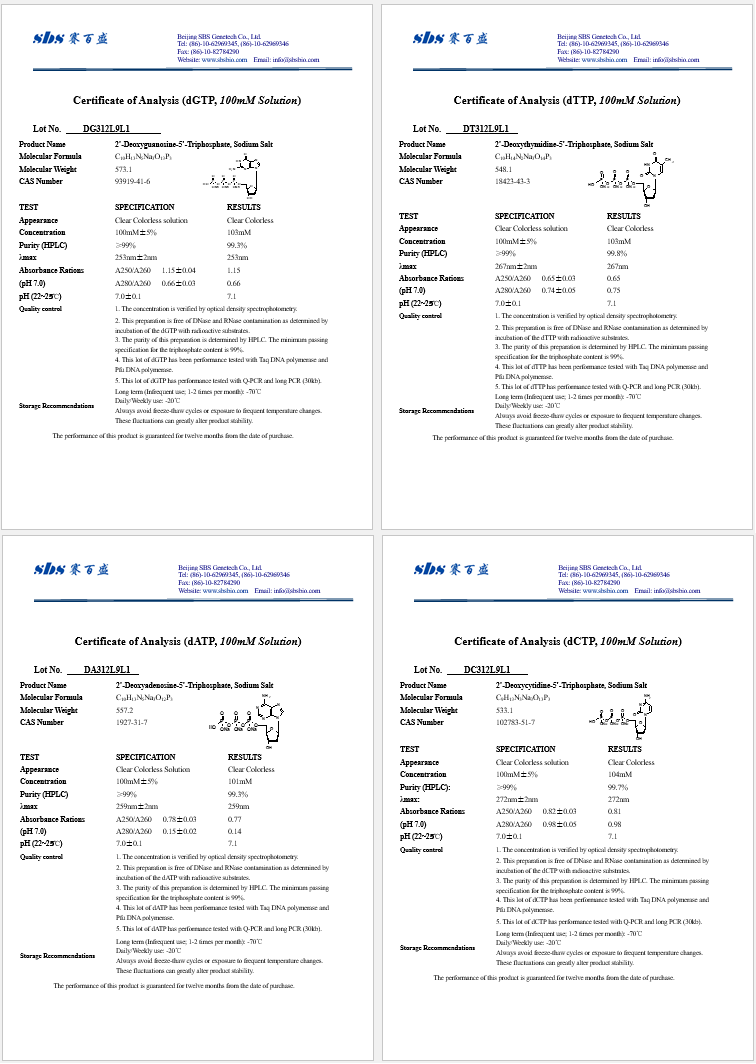
Analysis of certificate template for product
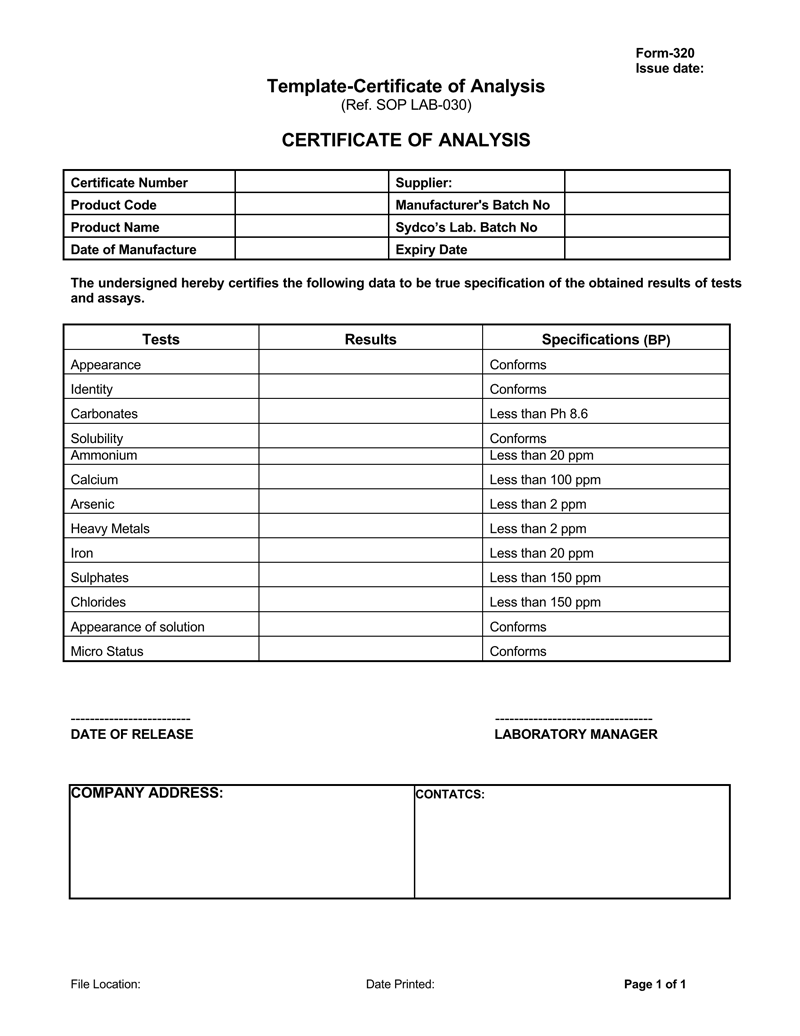
Sample certificate of analysis for drugs

Frequently Asked Questions
The difference between a Certificate of analysis and a Certificate of Conformance is that the latter is issued by a reliable authority confirming that the product follows the specifications and regulations without providing within the document the details about the inspection, the lab test results, or the conditions of the tests.
On the other hand, the certificate of Analysis will have complete transparency regarding the lab results and regulations. The document will contain all the details regarding the conditions of the tests, how it compares to a standard specification and the complete results of these tests.
The certificate of analysis is stored in PDF or Word document formats.











